Global ISO Certification Consultant Services – Qualitcert
ISO 13485 Certification Consulting Services in Surat
QUALIT
CERT
Qualitcert offers specialized ISO 13485 certification consulting services in Surat, focusing on helping organizations in the medical device industry develop and implement effective quality management systems. Their expert consultants provide tailored solutions to guide businesses through the entire certification process, including initial gap analysis, system design, implementation of quality management policies and procedures, and preparation for certification audits. Qualitcert’s approach ensures compliance with ISO 13485 standards by establishing processes for design, production, installation, and servicing of medical devices, as well as maintaining rigorous quality control and risk management practices. By partnering with Qualitcert, organizations in Surat can achieve ISO 13485 certification, demonstrating their commitment to producing safe and effective medical devices, meeting regulatory requirements, and ensuring customer satisfaction. This certification not only enhances product quality and operational efficiency but also boosts market access and competitiveness in the global medical device market. Qualitcert’s practical and systematic methodology ensures that businesses in Surat can effectively manage quality risks, improve product reliability, and achieve continuous improvement in their quality management systems.
ISO Certification Process – Step by Step Guide
The ISO certification process helps organizations implement international standards to improve quality, safety, efficiency, and compliance. Below is a structured step-by-step ISO certification process followed by professional ISO consultants and certification bodies.
ISO Application
The organization submits an application for ISO certification and defines the scope of certification including departments, processes, and operations.
Gap Analysis
ISO consultants analyze the current management system and identify gaps between existing processes and ISO standard requirements.
ISO Documentation
Preparation of ISO manuals, procedures, policies, risk assessments, and records required to comply with ISO standards.
System Implementation
ISO processes are implemented across departments with employee training, process control, and compliance monitoring.
Internal Audit
Internal auditors review the management system to verify compliance and identify corrective actions before the certification audit.
Management Review
Top management evaluates the effectiveness of the ISO management system and ensures readiness for certification.
Certification Audit
An accredited certification body conducts an external audit to verify compliance with ISO standards.
ISO Certification
After successful audit completion, the organization receives the official ISO certificate demonstrating compliance with international standards.
Surveillance Audits
Annual surveillance audits ensure continuous compliance and improvement of the ISO management system.
Please Reach Us Today

Please Reach Us Today


FAQ's
Frequently Asked Questions About ISO 13485 Certification in Surat
What is ISO 13485 Certification and why is it important for medical device companies in Surat?
How can I get ISO 13485 Certification in Surat with Qualitcert Certification Services?
What is the cost of ISO 13485 Certification consulting services in Surat?
How long does it take to get ISO 13485 Certification in Surat?
What documents are required for ISO 13485 Certification?
What are the benefits of ISO 13485 Certification for businesses in Surat?
Approach and Methodology used to implement Management System Standard
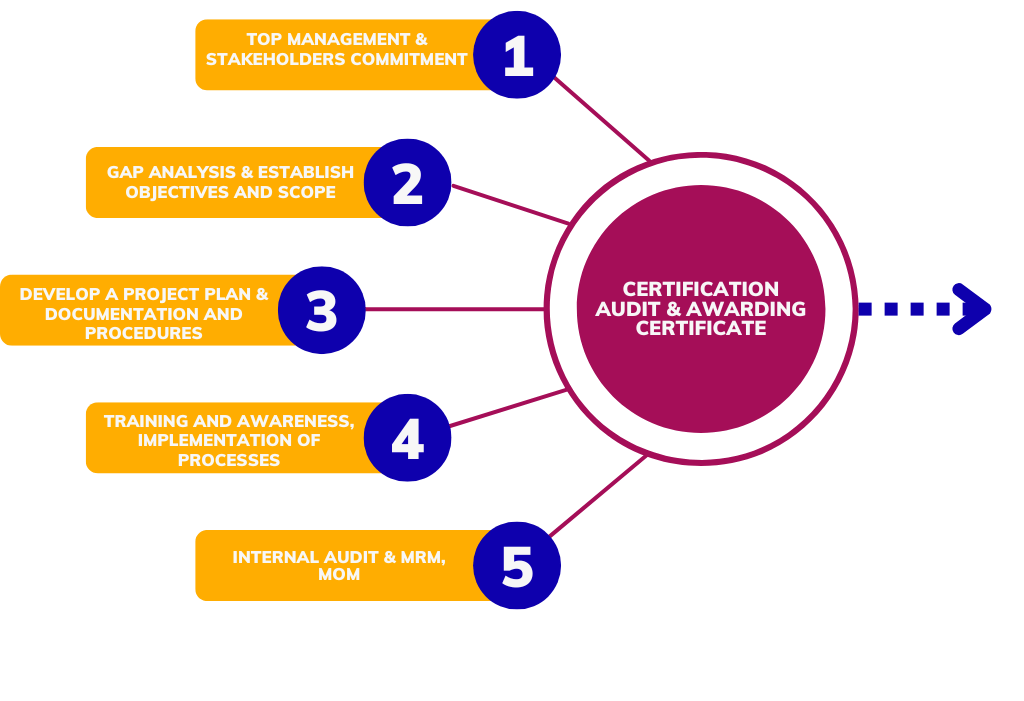
Implementing an ISO standards involves a structured methodology to ensure that the organization effectively meets the requirements of the chosen standard and achieves certification. Sometimes defined methodology may vary depending on factors such as the size of the organization, its industry, and the complexity of the ISO standard being implemented, the following steps provide a basic framework


OUR
Process
1, Determine the ISO Standard
2. Understand the Requirements
3. Training and Awareness
4. Implement the System
5. Internal Audit
6. Certification

Benefits of having ISO Certification
Enhanced Credibility and Reputation
Legal and Regulatory Compliance
Enhanced Customer Satisfaction
Access to Global Markets
Environmental Sustainability
Information Security
Our Achievements and Success
Our Clients





OUR
SERVICES
Our presence in Bangalore
Re-Design your system, create raving fan clients. Transform your business , Go Global
- ISO Certification in Peenya Bangalore
ISO Certification in Bommanahalli Bangalore
- ISO Certification in Whitefield Bangalore
- ISO Certification in Electronics City Bangalore
- ISO Certification in Bommasandra Bangalore
- ISO Certification in Rajajinagar Bangalore
- ISO Certification in Jigani Bangalore

