Global ISO Certification Consultant Services – Qualitcert
ISO 13485 Certification Consulting Services in Algeria
QUALIT
CERT
Qualitcert helps medical device companies create and maintain a strong Quality Management System (QMS) that conforms with ISO 13485 standards by offering specialized ISO 13485 certification & consulting services in Algeria. Their consulting approach begins with a thorough assessment to identify gaps in existing processes, followed by a tailored strategy to address these gaps, enhance regulatory compliance, and promote patient safety. Qualitcert’s expert consultants offer hands-on support in developing required documentation, conducting staff training, and integrating quality management practices into everyday operations. They also assist with internal audits and preparation for the final certification audit, ensuring full readiness for certification. By collaborating with Qualitcert, medical device companies in Algeria can achieve ISO 13485 certification, showcasing their commitment to quality, safety, and regulatory excellence while gaining a competitive edge in the healthcare industry.
Please Reach Us Today

Please Reach Us Today


Approach and Methodology used to implement Management System Standard
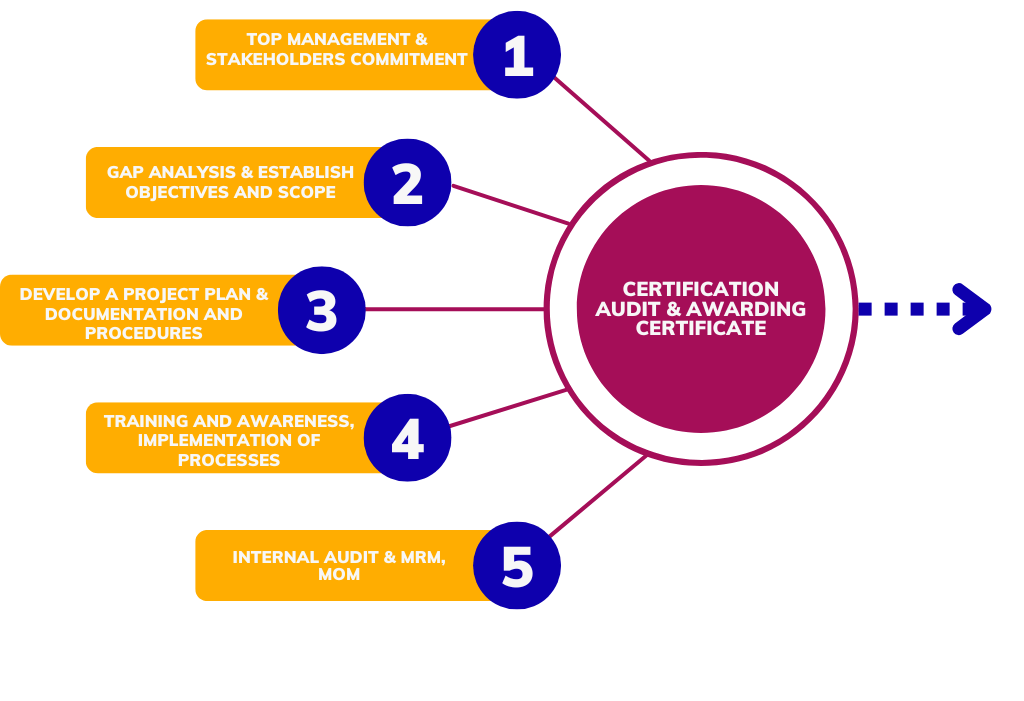
Implementing an ISO standards involves a structured methodology to ensure that the organization effectively meets the requirements of the chosen standard and achieves certification. Sometimes defined methodology may vary depending on factors such as the size of the organization, its industry, and the complexity of the ISO standard being implemented, the following steps provide a basic framework


OUR
Process
1, Determine the ISO Standard
2. Understand the Requirements
3. Training and Awareness
4. Implement the System
5. Internal Audit
6. Certification

Benefits of having ISO Certification
Enhanced Credibility and Reputation
Legal and Regulatory Compliance
Enhanced Customer Satisfaction
Access to Global Markets
Environmental Sustainability
Information Security
Our Achievements and Success
Our Clients





OUR
SERVICES
Our presence in Bangalore
Re-Design your system, create raving fan clients. Transform your business , Go Global
- ISO Certification in Peenya Bangalore
ISO Certification in Bommanahalli Bangalore
- ISO Certification in Whitefield Bangalore
- ISO Certification in Electronics City Bangalore
- ISO Certification in Bommasandra Bangalore
- ISO Certification in Rajajinagar Bangalore
- ISO Certification in Jigani Bangalore

